OSD App Clinical Trial Management
A comprehensive no-code application built with Google AppSheet to digitize, manage, and track the entire Operational Specifications Document (OSD) process for clinical trials, from study details to reporting.
The Problem
The management of clinical trial processes, particularly the Operational Specifications Document (OSD), is often complex, manual, and fragmented. This leads to inefficiencies, errors in data, difficulty in tracking progress, and challenges in communication among stakeholders. A centralized and digitized solution is needed to streamline these operations.
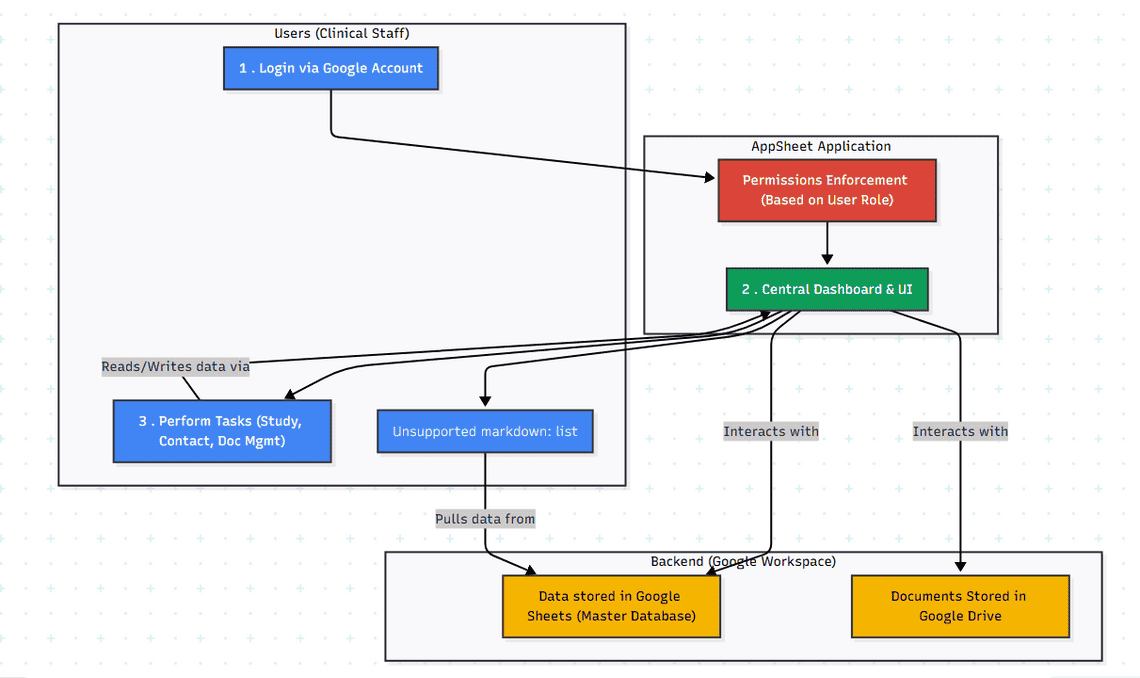
AI-Generated Diagram: Cross-Functional Flowchart for AppSheet Clinical Trial Management App
Workflow/User Journey
User Login: Users authenticate via Google accounts and are assigned roles based on their access level (Admin, Study Manager, Contact Manager, Viewer).
Study Management: Study Managers can create, edit, and view study details, including protocol information, timelines, and associated contacts.
Contact Management: Users can add, edit, and view contact information for individuals involved in studies, associating them with specific organizations and studies.
Workflow Tracking: Users can track the progress of OSD documents through various stages, such as data configuration, data flow, kit and supply management, test setup, reporting, and requisition setup.
Data Management: Users manage data related to tests, samples, results, and reference ranges.
Document Management: Users can store, track, and manage versions of OSD documents.
Reporting: Users can generate and view reports based on collected data, such as sample tracking, data analysis, and study progress.
Permissions Enforcement: The system ensures users only access data and functionalities relevant to their assigned role.
The Client/Target Audience
Clinical research organizations, pharmaceutical companies, academic institutions involved in clinical trials, study coordinators, project managers, data managers, regulatory affairs personnel, and other stakeholders responsible for managing the OSD process.
Technology Used
Google AppSheet
Google Sheets
Google Drive
Master Database Integration
No-Code Development
Mobile Application Development
Database Design
User Authentication
Role-Based Access Control
Data Security
Workflow Automation
Reporting & Analytics
Key Metrics/Achievements
Digitization of the entire OSD process, reducing reliance on manual documentation.
Improved accuracy and consistency of study and contact data by X%.
Enhanced visibility into workflow progress, leading to Y% faster completion of OSD stages.
Streamlined data management, reducing data entry errors by Z%.
Implementation of role-based permissions to ensure data security and user accountability.
Centralized platform for all OSD-related information, improving communication and collaboration.
Portfolios
Related Posts
Quick Links
Legal Stuff

